Abstract
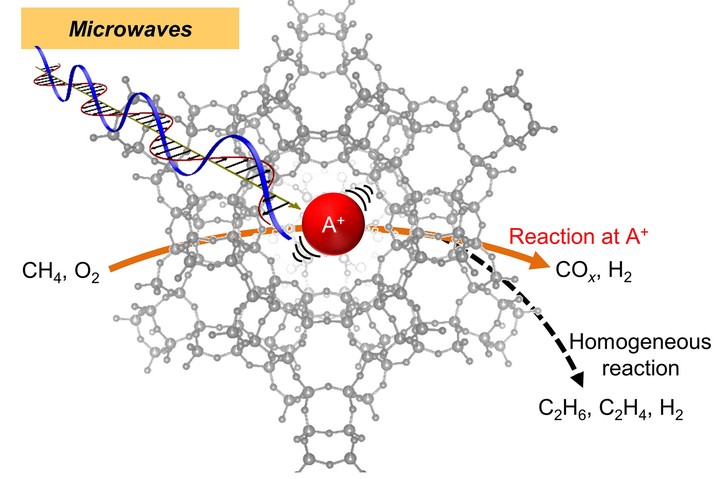
Microwave (MW)–driven catalytic systems are attracting attention not only as an aggressive electrification strategy of the chemical industry but also as creating a unique catalytic reaction field that conventional equilibrium heating cannot achieve. This study unlocked direct and selective heating of single alkali metal cations in the pores of aluminosilicate zeolites under MW. Selectively heated Cs$^+$ cations in FAU zeolite exhibited selective CH$_4$ combustion performance, that is, CO$_x$ generation at the heated Cs$^+$ cations selectively occurred while side reactions in the low-temperature gas phase were suppressed. The Cs$-$O pair distribution function revealed by synchrotron-based in situ x-ray total scattering gave us direct evidence of peculiar displacement induced by MW, which was consistent with the results of molecular dynamics simulation mimicking MW heating. The concept of selective monoatomic heating by MW is expected to open a next stage in “microwave catalysis” science by providing physicochemical insights into “microwave effects.”